One needs to know some basic properties of the given compound and its Lewis structure to understand its molecular geometry, polarity, and other such properties. SF4 is a chemical formula for Sulfur Tetrafluoride. It is a colorless corrosive gas that is used in the synthesis of several organofluorine compounds. SF4 is a rather hazardous compound but is used widely in chemical and pharmaceutical companies.
| Name of molecule | Sulfur Tetraflouride ( SF4) |
| No of Valence Electrons in the molecule | 34 |
| Hybridization of SF4 | sp3 hybridization |
| Bond Angles | 102 degrees and 173 degrees |
| Molecular Geometry of SF4 | Trigonal bipyramidal |
To understand this molecule’s properties, such as its reactivity, polarity, and more, one needs to know the SF4 Lewis structure first.
Contents
SF4 Molecular Geometry
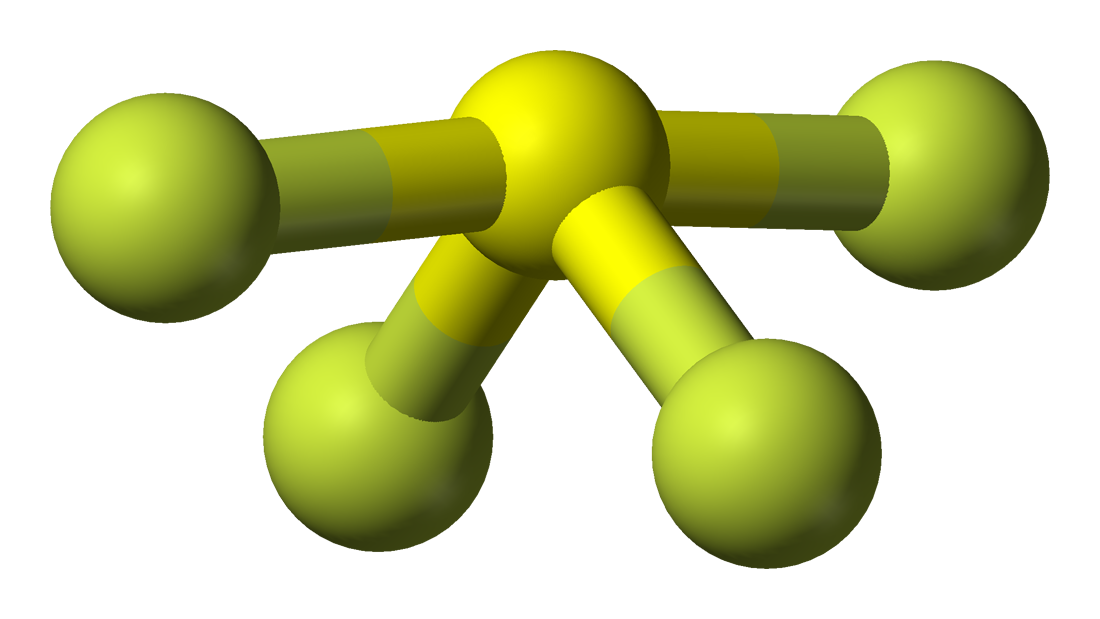
It is easy to understand the molecular geometry of a given molecule by using the molecular formula or VSEPR model. A molecular formula helps to know the exact number and type of atoms present in the given compound. Here there is one sulfur atom and four fluorine atoms in the compound, which makes it similar to the molecular formula of AX4E.
Molecules having a molecular formula of AX4E have trigonal bipyramidal molecular geometry. Here two fluorine atoms forming bonds with the sulfur atom are on the equatorial positions, and the rest two are on the axial positions. As there is one lone pair on the central atom, it repels the bonding pair of electrons, which tweaks the shape a little bit and makes it appear like a see-saw. The electrons follow this pattern of arrangement following the VSEPR rule to minimize the repulsion forces between the lone pairs of electrons to maximize the molecule’s stability.
Hence, SF4 has a trigonal bipyramidal molecular geometry.
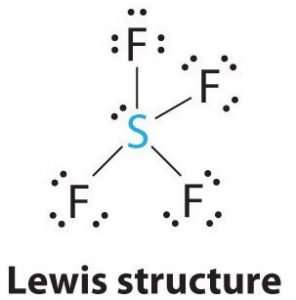
SF4 Lewis Structure
Lewis structure is a pictorial representation of the bonds and valence electrons in the molecule. The bonds formed between two atoms are depicted using lines, whereas the valence electrons not forming any bonds are shown by dots. The valence electrons that participate in forming bonds are called bonding pairs of electrons, whereas the electrons that do not participate or form any bonds are called nonbonding pairs of electrons or lone pairs.
And to draw the Lewis structure of SF4, we first need to know the total number of valence electrons in this molecule.
As one can probably see, there is one sulfur atom in this compound and four fluorine atoms. To know the total valence electrons of this compound, we need to know the valence electrons of both the atoms individually.
- Valence electrons of Sulfur: 6
- Valence electrons of Fluorine: 4* (7)
( as there are four fluorine atoms, we have to consider valence electrons of all atoms)
Total number of valence electrons in SF4 = number of valence electrons in sulfur + number of valence electrons in fluorine
= 6 + 28
= 34 valence electrons
Now that we know the total number of valence electrons, it would become easy for us to understand the bond formation between the atoms and the complete arrangement of the molecule too.
Sulfur will be the central atom in this molecule as it is the least electronegative, with four fluorine atoms forming bonds on the sides of this central atom. Every fluorine atom will form a bond with the central atom, which means there will be four bonds in the molecule structure using up four valence electrons of fluorine atoms and 4 electrons of the sulfur atom. So now, eight valence electrons are used, reducing the number of valence electrons from 34 to 24. All the fluorine atoms have six valence electrons, and the central atom has two valence electrons.
Draw lines between S and F to show bonds and for lone pairs of electrons, use dots. Each fluorine atom will have three pairs of 6 valence electrons ( shown as dots) on the atom, along with one bond with sulfur. In contrast, the central atom will have two valence electrons and four bonds.
Hence, the central atom, sulfur, will have one lone pair of electrons and four bonding pairs of electrons in the Lewis structure of SF4. At the same time, each fluorine atom will have three lone pairs.
Is SF4 polar?
Once we know the Lewis structure and molecular geometry of the given compound, it becomes easier to depict the molecule’s polarity. Here, one lone pair on the central sulfur atom and four bonding pairs of electrons leads to the asymmetric distribution of electrons on the central atom.
Also, as the shape of the molecule is like a see-saw, two fluorine atoms can cancel out each other’s dipole moment, but the rest two can’t due to the electrons’ arrangement. And as fluorine atoms are more electronegative than the sulfur atom, it results in uneven distribution of the charge. Hence the dipole moment is not canceled, which makes the molecule polar. So yes, SF4 is polar.
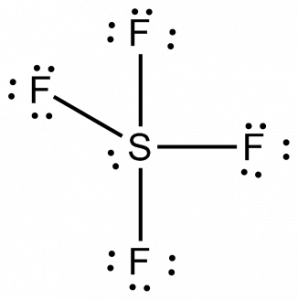
SF4 Hybridization
To know the hybridization of the SF4 molecule, let us first look at the regions of electron density for the central atom.
Sulfur has four bonding pairs of electrons and one lone pair, making its total number of regions for electron density 5. Hence the sulfur atom uses five hybridized orbitals, one 3s orbital, three 3p orbitals, and one 3d orbital. This arrangement of electrons around the atom and hybridized orbitals leads to the sp3d hybridization. One can also use the steric number to know the hybridization; here, the steric number is 5 for the sulfur atom.
Thus SF4 has sp3d hybridization.
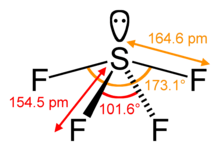
SF4 Bond angles and shape
The central sulfur atom forms four bonds with the neighboring fluorine atoms and has one lone pair of electrons. Fluorine atoms on the equatorial positions have the bond angles of 102 degrees, and the axial ones have 173 degrees, which are a little different than the trigonal bipyramidal molecular geometry leading to a see-saw shape.
The lone pair on the central atom leads to the change in the bond angles from 120 degrees to 102 degrees for equatorial fluorine atoms and 173 degrees instead of 180 degrees for axial fluorine atoms.
Concluding Remarks
To conclude all the properties we can say that,
- Sulfur Tetrafluoride has 34 valence electrons, out of which it forms four covalent bonds and one lone pair of electrons on the central atom in its Lewis structure.
- There are three lone pairs on each fluorine atom.
- It has a molecular geometry of the formula AX4E; it forms a see-saw shape and has a trigonal bipyramidal molecular geometry.
- SF4 has sp3d hybridization and is polar in nature.



Amazing Explanation!!! Thanks a lot for helping out..
GOOD
GOOD:)
Is sf4 is planar
Nice explanation
Thanks for your article. Hunting accurate information is among the biggest
issues for the younger generation.
But what about drago’s rule.
Isn’t drago’s rule being violated in your answer?
Please reply ,i am very much confused.
Why is not forming a see saw shape with 90° and 120° bond Angles