Polarity is one of those properties that impact all the other physical properties that include its boiling point, melting point, and even solubility. The property is essential to know all the other chemical properties and their uses and limitations of using.
But before diving into the process of how to tell if a molecule is polar or not, let us quickly go through the definition of polarity for your better understanding.
Polarity: It is a separation of electric charges in the molecule due to the electric dipole moment. Such separation of charges leads to the positively charged end of a molecule and the negatively charged end. These two polar ends act as electric poles that differ in electric charges. This property of the molecule having different electric charges is termed polarity.
Depending upon the electric charges of the molecule, it is classified as polar or nonpolar. To know how they exactly differ from each other, check out these differences between polar and nonpolar molecules.
Contents
Polar vs Nonpolar molecules
| Polar molecules | Non-polar molecules |
| The molecules having positively charged end and negatively charged end due to the difference in the charges of atoms present in the molecules are polar molecules. | The molecules that do not have such separation of electric charges are nonpolar. |
| Most of the polar molecules have an asymmetric or uneven distribution of electrons. | Nonpolar molecules have a symmetrical distribution of electrons. |
| When highly electronegative atom bonds with a relatively less electronegative atom, polar molecules are formed. | Nonpolar molecules are formed when there is quite a little difference in the electronegativities of the atoms forming bonds in the molecule. |
| Due to the separation of electric charges, there is a net dipole moment in the polar molecules. | As there is no separation of electric charges, there is no net dipole moment in nonpolar molecules. |
| These molecules react with polar molecules to form solutions. | Nonpolar molecules do not have such reactivity. |
| Polar molecules dissolve in polar molecules only. | Nonpolar molecules also dissolve in nonpolar molecules only. |
| Water is a polar molecule as there is a difference in electronegativities of Oxygen and Hydrogen atoms. | All hydrocarbons are mostly nonpolar. |
Examples of polar and nonpolar molecules
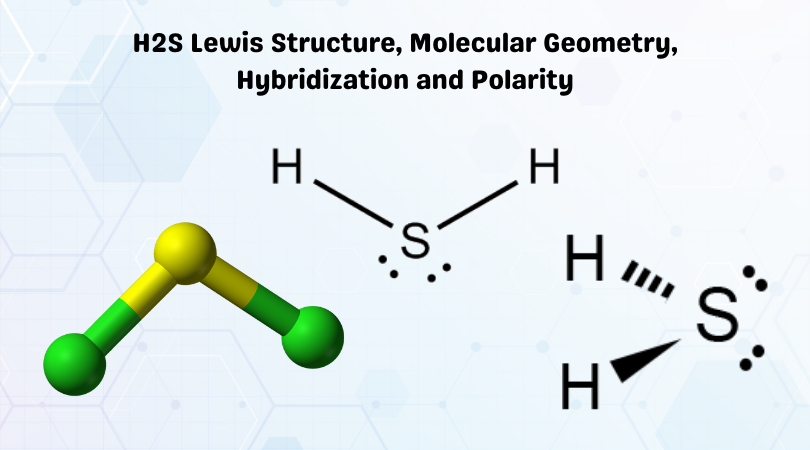
Polar molecules: Water, alcohol, Sulphur dioxide, ammonia, ethanol, hydrogen sulfide, bent molecules (those with a significant bond angle) in general.
Non-polar molecules: Hydrocarbons (gasoline, toluene), homo-nuclear diatomic molecules (O2, N2, Cl2, H2, etc.), noble gases, benzene, methane, ethylene, carbon tetrachloride
How to Determine if a Molecule is Polar Or Nonpolar
- Start by drawing its Lewis structure. This rule applies to all molecules except hydrocarbons and molecules with two atoms of the same element.
- The Lewis structure will help you analyze the shape of the molecule given to you
- Determine which of the five categories of shapes your molecule falls into linear, tetrahedral, trigonal planar, bent, trigonal pyramid. The first three are symmetric shapes and the last two are asymmetric shapes.
- As learned before, non-polar molecules are perfectly symmetrical while polar molecules are not. This means that if the shape of the molecule given to you is a bent or trigonal pyramid, it is a polar molecule.
- Remember that asymmetry applies even if the outer atoms are the same. The arrangement of the atoms matters more.
- Now, let’s dissect the symmetric molecules. All the atoms that are attached to the central atom must be the same if it is a nonpolar molecule. If different kinds of atoms are attached to the central atom, the molecule is polar.
Are all bent molecules polar?
Mostly, yes. As aforesaid, bent molecules are asymmetrical just like trigonal pyramids and that means that they are polar molecules.
Examples of bent molecules are H2O, NO2, CH2, and SCl2.
The exceptions
There are a few exceptions to the rules of polar and non-polar molecules and the C-H bond is a classic example. This molecule is nonpolar even though the bonds are slightly polar.
NCl3 is a rare example. Nitrogen and chlorine are both electronegative. That’s why their bond (N-Cl) is non-polar. However, when you see the NCl3 molecule you will see that the nitrogen atom has a single pair of electrons. This makes the molecule polar by nature.
SO3 and BH3 are other examples. They have polar bonds but they are non-polar in nature. O3, on the other hand, has a nonpolar bond but is polar by nature.
We hope you will now know how to tell if a molecule is polar or nonpolar. If you have any insights or questions, we would love to answer them for you!



Given information was extremely useful for me. I appreciated. Thank you so much.
Thanks, comprehensible
Thank you so much! You broke it down into steps that I can understand.
Always happy to help!