Hydrogen Sulfide is a common chemical compound that is useful for analyzing inorganic compounds of metal ions. It has the chemical formula of H2S. The molecule has two Hydrogen atoms and a single Sulfur atom. H2S is also a precursor for elemental Sulfur. It also plays a vital role in signaling pathways in the human body. So to understand the hybridization, polarity, and molecular geometry of this compound, it is essential to know its Lewis structure.
| Name of molecule | Hydrogen Disulfide ( H2S) |
| No of Valence Electrons in the molecule | 8 |
| Hybridization of CO2 | sp3 hybridization |
| Bond Angles | 104.5 degrees |
| Molecular Geometry of CO2 | Bent |
Before knowing its Lewis structure, let us calculate the total number of valence electrons in Hydrogen Sulfide as these electrons participate in bond formation and help us study Lewis structure with ease.
Contents
Valence electrons of H2S
To know the total number of valence electrons in Hydrogen Sulfide we need to add the valence electrons of both Hydrogen and Sulfur atoms. There are two atoms of Hydrogen and a single atom of Sulfur in the compound.
Each Hydrogen atom has only one electron which is also its valence electron
Hence there are two valence electrons for Hydrogen atom ( as there are two Hydrogen atoms)
Sulfur has six valence electrons.
Total number of valence electrons in H2S
= 2(1) + 6
= 8
Thus, there are a total of eight valence electrons in H2S.
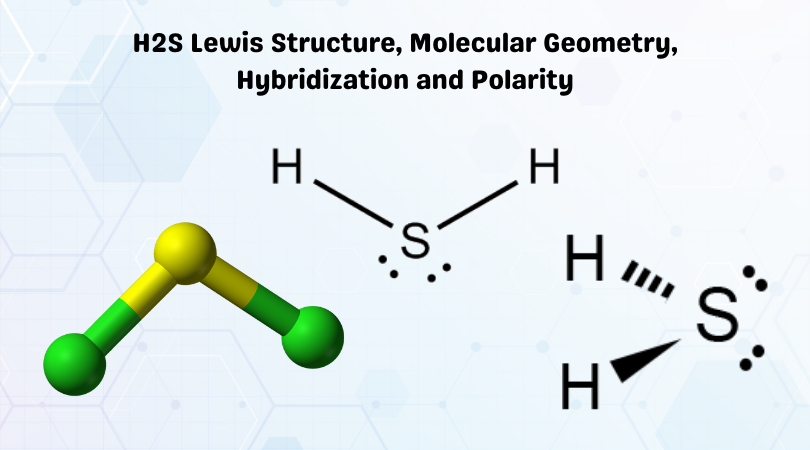
H2S Lewis Structure
The Lewis structure of any compound is a structural representation of the valence electrons participating in the formation of bond along with the nonbonding electron pairs. Knowing the Lewis structure of a given chemical compound is essential as it provides the necessary information about all other chemical properties of the compound.
The representation is displayed using dots and lines that represent electrons. Dots represent the electrons that do not participate in the bond formation. Whereas, the lines represent the bonds in the compound. The structure is made based on the Octet Rule. The Octet Rule of chemistry states that there should be eight electrons in the outer shell of an element for it to be stable.
The Lewis Structure of Hydrogen Sulfide is easy to draw and understand. In this compound, both the hydrogen atoms require one electron to make the covalent bond with Sulfur. The Lewis structure of H2S is similar to H2S. Sulfur needs eight electrons to fulfill the requirements for Octet Rule. But Hydrogen only requires a single electron to become stable as it belongs to Group 1 elements.
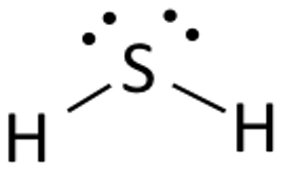
Place the Sulphur atom in the middle and arrange its valence electrons around it.
Now place two Hydrogen atoms on both sides of the central atom.
Here the valence electrons of Sulfur are used to make Hydrogen stable by donating an electron to both the atoms of Hydrogen. Two out of six valence electrons participate in bond formation.
Draw lines ( for showing bonds) between Hydrogen and Sulphur atoms.
The remaining four electrons are nonbonding electrons and are hence displayed as dots near the Sulfur atom. In total there are two pairs of non-bonding electrons in this compound and Hydrogen atoms have a complete valence shell.
H2S Hybridization
To understand the hybridization of H2S, it is vital to know two things first:
- The number of bonds in the compound and its type
It is essential to know the type of bonding in the compound to know its hybridization. In the H2S molecule, two Hydrogen atoms form a bond with the central Sulfur atom. Two single bonds are formed in the molecule. These bonds take up four valence electrons, and hence there are four other valence electrons left. While forming a bond the s orbital of the Hydrogen atom overlaps with p orbital of the Sulfur atom. The lone pair of electrons takes up two of the sp3 orbitals. The other two orbitals of sp3 overlap with 1s orbital of the Hydrogen atom.
This arrangement leads to the sp3 hybridization of H2S molecule. The overlapping of the two-hybrid orbitals forms two sigma bonds between Hydrogen and Sulfur atom.
- The steric number of the given compound
The steric number is defined as the region of electron density seen around the central atom. In this case, as Sulfur is the central atom the steric number of the compound will be the cumulative total of atoms bonded to the central atom and the number of lone pairs of electrons around the central atom.
The formula for finding this number is:
Steric Number (SN)
= No of sigma bonds on the central atom +No of pi lone pairs on the central atom
In this case of H2S molecules, there are two sigma bonds, and there are two lone pairs of electrons on the central atom.

Thus SN of H2S molecule = 2+2
=4
As the Steric number of H2S is four, it has two
hybrid orbitals and two lone pairs of electrons that make it an sp3 hybridization.
Now that we know the Lewis structure and hybridization of the molecule, it is easy to determine its molecular geometry.
H2S Molecular geometry
Hybridization of the given molecule H2S is sp3; the Sulfur atom is in center bonding with two Hydrogen atoms forming the bond angle less than 180 degrees. According to the VSEPR theory, the lone pairs of electrons repel each other, but as the Sulfur atom is less electronegative, the bond angle decreases to 104.5 degrees.
This decrease in the angle leads to a bent structure.
Another way to know Molecular geometry is by the AXN method. Where A denotes the central atom, X is for the number bonded pair of electrons and N is for the lone pair of electrons. Here the general formula will be AX2N2 due to two bonding pairs and two lone pairs of electrons. Thus this molecule shows a bent geometry.
H2S Polarity
Now that we have the required information about the molecule, it is easy to determine its polarity. The polarity of any given molecule depends on its molecular geometry and the overall electronegativity. As the Sulfur atom is less electronegative, the overall electronegativity of the compound is less than 0.4, which makes it nonpolar.
Thus it can be said that the H2S molecule is nonpolar.
Concluding remarks
From the above information, it can be concluded that the H2S molecule has eight valence electrons, sp3 hybridization, bent molecular geometry, and is nonpolar.


Sulfur has 2p orbital which overlaps with Hydrogen’s 1s orbital, therefore due to huge difference in energy, 2s orbital would not get hybridized since this process can only occur when the two overlapping orbitals are of comparable energy. But you have mentioned in the article that it is sp3 hybridized, can you please give an explanation according to my statement?
H2s is drago molecule….it doesnt show hybridisation ??♀ How on earth can u write its hybdzn to be sp3??♀
Thank you.
i think you mean 3p but yes due to huge gap in energy they cant hybridize. Bonding is purely P orbital based. dragos rule is applicable