Carbon Dioxide is one of the best compounds to start with learning the concepts of Lewis structure and Molecular Geometry. This molecule can be a good start for beginners who want to learn the fundamentals of such concepts and want to know how to draw Lewis dot structures for other molecules as well.
CO2 or Carbon Dioxide is made up of two types of atoms: Carbon and Oxygen. Although this gaseous molecule is known for its contribution to the greenhouse effect and global warming, one cannot deny that there are quite a lot of uses for this gas in several industries.
To understand the physical properties, reactivity, and other chemical properties of a given compound, it is essential to know its molecular geometry. And to help you understand it, I have discussed the CO2 Lewis structure and its hybridization below.
| Name of molecule | Carbon Dioxide ( CO2) |
| No of Valence Electrons in the molecule | 16 |
| Hybridization of CO2 | sp hybridization |
| Bond Angles | 180 degrees |
| Molecular Geometry of CO2 | Linear |
CO2 Lewis Structure
One needs to know the Lewis structure in order to understand the molecular geometry of any given molecule. This structure helps in knowing the arrangement of electrons in the molecules and the shape of the molecule. To know the lewis structure of CO2, one should first understand what precisely the Lewis structure is.
Lewis dot structure is a pictorial representation of the arrangement of the valence shell electrons in the molecule. These valence electrons are represented by drawing dots around the individual atoms, hence the Lewis dot structure. Drawing lines represent the bonds formed in the molecule.
Such structure helps in understanding the arrangement of atoms along with the electrons participating in the bond formation. Now that you know how the Lewis structure is drawn and its uses let us quickly look at the CO2 Lewis structure.
In CO2, the Carbon atom is in the central position as it is the least electronegative atom in the molecule. Two Oxygen atoms are located on the terminals where both these atoms share electrons and form bonds with the central Carbon atom.
To know the bond formation and the arrangement, let’s go through the valence electrons of all the atoms in the molecule.
- Valence electrons in Carbon: 4
- Valence electrons in Oxygen: 6*2 = 12 ( as there are two Oxygen atoms in the molecule, we will multiply it by 2)
Total number of valence electrons in the molecule = 16
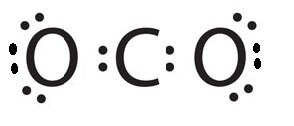
So, for now, place Carbon in the center position and draw four dots around it. Along with the place, two Oxygen atoms on both sides of the atom and draw six dots around each atom to represent their valence electrons.
You might know that a molecule needs to complete its octet to become stable and inactive by achieving an electronic configuration similar to the inert gases. This is done by either donating an electron or by accepting an electron. Here as the Oxygen atoms are more electronegative than the Carbon atom, the Carbon atom will donate its electrons to both these Oxygen atoms.

Now, as two oxygen atoms need two electrons each to complete their octets, it will share two electrons from the Carbon atom and form double bonds. Hence each Oxygen atom will form a double bond with the central atom.
So now draw two parallel lines between Oxygen atoms and Carbon atoms to show double bonds between the atoms. For Lewis structure of CO2, you will now have two Oxygen atoms forming double bonds with a Carbon atom.

As all the valence electrons of all the atoms are used, there are no lone pairs of electrons or non-bonding pairs of electrons in the molecule.
To further understand the molecular geometry of CO2, let us quickly go through its hybridization and bond angles as it will make it easy for us to understand the geometry.
CO2 Hybridization
The electronic configuration of the Carbon atom in its ground state is 1s22s22p2, and that of an Oxygen atom is 1s22s2p4. When the electrons are in an excited state, they jump to other orbitals.
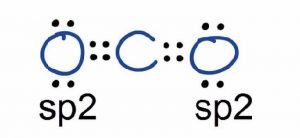
In its excited state, the atom’s electronic configuration becomes 1s2 2s1 2p3, so now every p-orbital of the atoms has one electron each. Here the 2s orbitals and one of the p-orbitals will hybridize to form 2 sp orbitals. In contrast, the Oxygen atom hybridizes to form three sp2 hybrid orbitals.
These two hybridized orbitals overlap with the two p-orbitals of the Oxygen atom that results in the formation of sigma bonds. Remaining electrons in the p-orbitals in the Oxygen atom form pi bonds.
As sp orbitals are hybridized to form the bonds, CO2 has an sp hybridization.
CO2 Molecular Geometry
The molecular Geometry of any compound is based on the arrangement of atoms, electron pairs, and bonds. Here in CO2, both Oxygen atoms form sigma bonds with the central carbon atom and complete their octet. As a result, there are no lone pairs of electrons, but bonding pairs of electrons also repel each other. Due to these repulsive forces between the valence shell electron pairs, the CO2 molecule acquires a linear shape to keep the repulsion at the least.
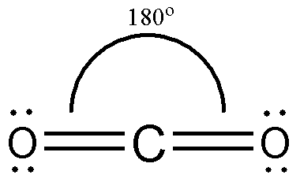
Hence CO2 has a linear molecular geometry with the bond angles of 180 degrees and symmetric distribution of electrons.
Summary
To summarize this blog, we can say that Carbon Dioxide has a linear molecular geometry. It has an sp hybridization and has bond angles of 180 degrees. There are no lone pairs of electrons in the molecule, and there is a symmetric distribution of the electrons in its structure. Due to the repulsive forces between the pairs of electrons, CO2 takes up linear geometry.


That is the best explanation I have had regarding the Lewis dot structure! I am no longer confused about his. Thank you very much.
I like the teaching method of arranging the Carbon in the center, then the oxygen atoms, then the inner bonds, the outer, and filling in the Carbon valence shell. A very “building block”, logical and pedagogical approach.
Thanks