The chemical formula N3– represents the Azide ion. The Azide ion is a conjugate base of Hydrazoic acid (HN3). It is composed of three Nitrogen atoms and can have multiple resonance structures.
Azides behave similarly to halogen-based compounds in that they react rapidly with other substances by displacement of the Azide group. This process gives rise to many types of substances.
One of the most common Azide-based substances is Sodium Azide (NaN3). Tons of this compound are produced annually to be used in the automobile industry (airbags), as biocides, and for organic synthesis of amine in the laboratory.
Other Azides such as Pb(N3)2 (Lead Azide) are used in construction as detonators. This is because most Azides are unstable and are highly prone to shock.
Most Azides can be derived from Sodium Azide. Aliphatic compounds undergo nucleophilic substitution, while acyl azides can be obtained by substitution with acyl chlorides.
Azide compounds are highly toxic, with Sodium Azide being on the same level of toxicity as alkali-based cyanide compounds. They have proven to be fatal and must be handled and disposed of with care.
The Lewis and corresponding resonance structures provide a good deal of information about the Azide anion’s properties. Some of them are listed below:
| Name of the ion | Azide (N3–) |
| No. of valence electrons | (5 x 3) + 1 = 16 valence electrons |
| Hybridization of the central atom | sp |
| Bond Angles | 180° |
| Molecular Geometry of N3– | Linear Molecular Geometry |
Contents
N3- Valence Electrons
Lewis structures use valence electrons to represent chemical bonds between elements. Valence electrons are found in the outermost shells of the atom, where the force of attraction from the center is the weakest.
This makes the valence electrons susceptible to excitation, and therefore, they can break away to help form chemical bonds.
The Azide anion comprises three Nitrogen atoms. The valence electrons from these atoms help form the Azide anion Lewis structure.
N3– comprises three Nitrogen atoms that all contribute valence electrons based on their position in the periodic table of elements.
Nitrogen is in group 5 of the periodic table with the electronic configuration 1s22s22p3.Therefore, the three Nitrogen atoms contribute 5 x 3 = 15 valence electrons.
Since the Azide ion is an anion, it’s a negative charge of -1. This negative charge contributes one valence electron as well.
Therefore, the total number of valence electrons in the Azide anion [N3–] is given by:
15[N] + 1[Neg-charge] = 16 valence electrons.
N3– Lewis Structure
Lewis structures are a schematic representation of molecules and their constituent chemical bonds. They help give insight into a molecule’s geometry, shape, and polarity, among other data.
The number of valence electrons available is the first step towards the Lewis structure. This has been discussed in the previous section. Next, a skeletal structure is arranged. The three Nitrogen atoms available are placed adjacent to one another. This is a homogenous ion with three atoms belonging to the same element.
We will now start placing the valence electrons to form chemical bonds and fulfill the octet requirements. Two electrons are placed between the atoms to form the chemical bonds, as shown below.

The remaining valence electrons are placed around the atoms to fill their outermost shells. The two outer Nitrogen atoms are filled. However, the central Nitrogen atom, having only four valence electrons, needs to form bonds with the adjacent atoms to attain its octet.

This predicament is overcome by the formation of chemical bonds between the central Nitrogen atom and its adjacent Nitrogen atoms. This is shown below:

The above arrangement is stable as all of the octet requirements have been fulfilled. The final Lewis structure is shown below:
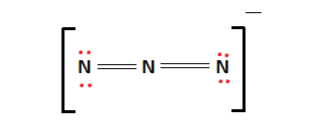
There are resonance structures that can be derived for the azide ion. These are shown below.

In the above resonance structures, the presence of triple bonds and the lack of an octet present challenges to the ion’s stability. A double charge on a small atom-like Nitrogen is unlikely in the case of the triple bonded resonance structures.
In the latter two, the lack of an octet leads to relative instability. As such, the Lewis structure shown above with double bonds and a complete octet is the one that we shall consider.
N3– Hybridization
To determine the hybridization of the central atom in the Azide ion, it is pertinent that we observe its Lewis structure discussed above.
The central Nitrogen atom is chemically bonded to two adjacent Nitrogen atoms through double bonds. As we’ve discussed the concept of electron regions earlier, we can quickly determine the hybridization from this data.
Two regions are surrounding the central Nitrogen atom. Therefore, the hybridization of the Azide ion is determined to sp.
N3– Bond Angles
The Nitrogen atoms present will repel each other in accordance with the VSEPR theory, arranging themselves in a linear manner. This leads to bond angles of 180°.
N3– Molecular Geometry
Observing the Lewis structure of the compound gives us insight into the molecular geometry and electronic shape of a particular compound.
The Azide Lewis structure comprises three Nitrogen atoms. The central Nitrogen atom forms two double bonds with the adjacent nitrogen atoms. According to the VSEPR theory, the atoms will repel each other to give a Linear Geometry.
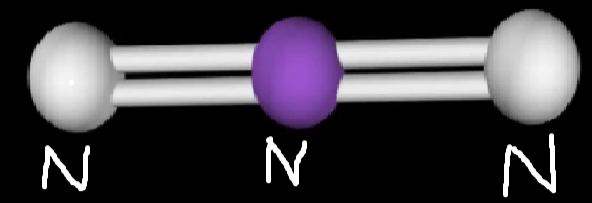
This can be confirmed with the A-X-N method.
‘A’ here represents the central atom Nitrogen. Therefore, ‘A’ = 1.
‘X’ represents the number of atoms bonded to the central atom. In this case, two more Nitrogen atoms are bonded to the central nitrogen atom.
Therefore, X =2.
‘N’ represents the number of lone pairs attached to the central atom. In this case, N = 0 as there are no lone pairs.
Therefore, that would give us AX2 for the Azide ion (N3–)
From the A-X-N table below, we can determine the molecular geometry for N3–.
| Formula | Shape | Bond Angle (Theoretical) |
| AX2 | Linear | 180 |
| AX3 | Trigonal Planar | 120 |
| AX4 | Tetrahedral | 109.5 |
| AX5 | Trigonal Bipyrimidal | 120, 90 |
| AX6 | Octahedral | 90 |
| AX2N | Bent | 120 |
| AX2N2 | Bent | 109.5 |
From the above table, it can be observed that an AX2 arrangement corresponds to a Linear Molecular geometry.
CONCLUDING REMARKS
Let’s quickly summarize the salient features of N3–
- The N3– ion comprises three atoms, with all of them being Nitrogen. One of the Nitrogen atoms takes its place in the center.
- The central Nitrogen atom forms double bonds with its neighbors in order to attain its octet.
- The hybridization of the Azide ion is sp.
- N3- has a Linear molecular geometry. This results in bond angles of 180°.

Nice article .f
Your work should really be appreciated as it helps lots of science students like me to master their concepts…
Thank you
Always happy to help, Aaliya!