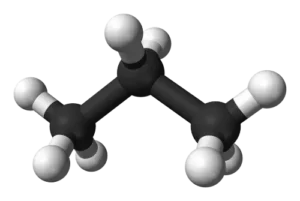
The chemical formula C3H8 represents the Propane molecule. The substance is a colorless, odorless gas at standard temperature and pressure. Due to a low boiling point of -42.1° C, propane can easily be liquefied at elevated pressure rates. This makes it feasible for transportation and other applications.
Propane is sourced as a by-product from the isolation of alkanes in natural gas processing and the cracking of petroleum in its refining process. In cracking, long-chain hydrocarbons are broken down into lighter ones by breaking the C-C bond. Millions of barrels of propane are then transported to Salt Caverns for storage purposes.
Propane burns in oxygen to release heat alongside water and carbon dioxide like other alkanes.
An insufficient oxygen supply results in Carbon Monoxide (CO) and soot(carbon) products.
In comparison to coal, propane has a much cleaner burn; and due to its high hydrogen content, it burns hotter than diesel. Trucks fueled by diesel engines use propane boosts to improve efficiency by at least 20%.
Propane is widely used as a portable energy source and lends itself to many applications. Some of these include it being used as a refrigerant, as soldering gas, in hot-air balloons, etc. Propane is highly flammable, however, and must be handled with care.
The polarity of the Propane molecule can be determined by examining its structure and understanding the nature of the chemical bonds present.
Contents
Electronegativity and Bond Nature
A difference in electronegativity generates regions of electropositive and electronegative in the ion or molecule. This, in turn, has an add-on effect on dipole moments and attraction, in turn affecting polarity. Calculating this difference will also tell us about the nature of the chemical bond in place.
We look to the periodic table to determine the difference in charges between the Carbon and Hydrogen atoms. In this case the difference is 2.5 [C] – 2.20 [H] = 0.3
Now, we look to the Pauling scale to help determine bond nature and polarity. According to the Pauling scale, if the difference in electronegativity lies between 0.5 and 2.0, the bonds are polar by nature.
In this case, the Carbon-Hydrogen bonds are sufficiently covalent, while the Carbon-Carbon bonds are covalent as well. This is indicative of a non-polar nature.
Molecular Shape and Symmetry
The propane molecule comprises 3 Carbon atoms and 8 Hydrogen atoms. The Lewis structure of the propane atom along with its molecular geometry is shown below:
It is observed that the structure has no lone pairs and is entirely symmetrical. This is also indicative of a non-polar nature.
Net Dipole Moment
Upon observing the molecular geometry of propane, we find that it possesses no lone pairs and that it is symmetrical in structure. This means that there are no dipoles present in the C3H8 molecule. This means that there is no net dipole moment.
Conclusion
As discussed above, the three properties that help determine chemical polarity all indicate that the propane molecule is non-polar. The difference in electronegativity is less than 0.5, in this case, 0.3. Furthermore, the molecular geometry is symmetrical and possesses no lone pairs.
This, in turn, means that there is no net dipole moment.
Therefore, the propane molecule is non-polar.