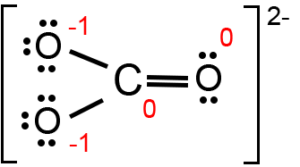
The chemical formula CO32- represents the Carbonate ion. Carbonates belong to both the organic and inorganic class. Inorganic Carbonates are derived from Carbonic acid (formed when CO2 dissolves in water).
They exist as salts of Carbonic acid and as ions of Sodium and Calcium among other metals. Carbonates are prominent in nature and comprise minerals such as Limestone and shells of polyps and molluscs.
We say that the Carbonate ion is non-polar. Let’s see why that is the case.
To determine the polarity of the CO32- ion, we must first account for its properties. These include its electronegativity, its molecular geometry and its resulting dipole moment, if any.
Contents
Electronegativity and Bond Nature
To determine if the bonds present in the CO32- ion are polar or non-polar, we look to the periodic table. The difference in charges between the Oxygen and Carbon atoms is 0.89.
Now, in accordance with the Pauling scale, this tells us that the Oxygen-Carbon bond in the CO32- ion is polar in nature.
Molecular Shape and Symmetry
The CO32- ion has three Oxygen atoms bonded to the central Carbon atom as shown in the figure.

It has three resonance structures as the double bond between the Oxygen atom and Carbon can be placed between any of the other Oxygen atoms as well.
According to the VSEPR theory, the Oxygen atoms repel each other and spread far away in an even manner. This results in equivalent bond angles of 120°, making the structure symmetrical in nature. It has a Trigonal Planar geometry.
Net Dipole Moment and 2- Charge
As shown above, the CO32- ion has symmetrical nature. Therefore, its polar bonds are distributed evenly. This results in the induced charges being cancelled out resulting in a zero net dipole moment.
Dipoles are in opposite directions as shown in the figure.

We know that the CO32- ion has a negative 2 charge. This must mean it’s polar, right?
Well, that’s not the case. Due to its Trigonal Planar structure which is symmetrical, the negative 2 charge distributes itself evenly over the three oxygen molecules as shown in the figure.
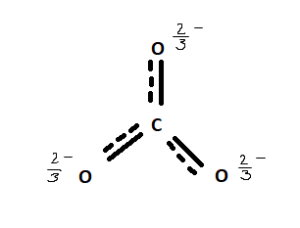
So, CO32- Polar or Nonpolar?
As shown above, the CO32- ion has polar bonds. However, these bonds and their constituent negative charge are distributed evenly due to their symmetric shape. This results in a zero net dipole moment. Therefore, the CO32- ion is non-polar in nature.