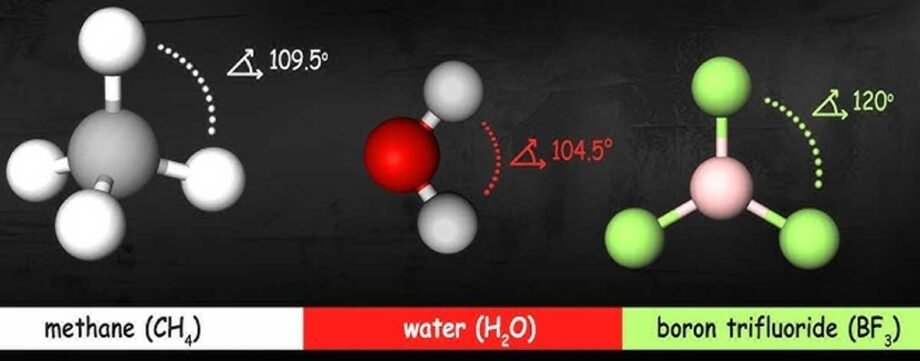
Molecular geometry is the three-dimensional arrangement of atoms in a molecule that determines how a material behaves under force, heat, and electricity.
That atomic-scale structure may seem distant from a factory floor, yet it directly decides whether a bracket bends smoothly, a weld holds under stress, or a housing resists long-term creep.
Global manufacturing contributes trillions of dollars in value each year, and even small improvements in material effectiveness can scale into a massive economic impact.
At the same time, advanced industries such as energy, electronics, and automation are demanding tighter tolerances and higher durability, which puts atomic structure under real pressure.
In this guide, you will see how atomic bonds influence mechanical strength, how crystal structures shape ductility, which manufacturing processes rely on microstructure, and how motion control and forming equipment depend on predictable material traits.
By the end, the link between chemistry and industrial performance will feel practical rather than theoretical.
Contents
What Is Molecular Geometry and Why Does It Matter in Manufacturing?
Molecular geometry is the three-dimensional shape of a molecule that governs how atoms share electrons and interact with one another.
In manufacturing, that shape becomes stiffness, toughness, conductivity, and thermal stability.
When a polymer enclosure softens too early or a metal cracks at a bend, the root cause is often structural.
Bond angles, electron distribution, and intermolecular bonds define how tightly atoms pack and how easily they move under stress.
A tightly packed structure can resist deformation but may fail abruptly.
A more flexible structure can absorb energy but may deform permanently under load.
Manufacturing processes such as bending, machining, and welding impose localized tension and heat.
If the internal geometry cannot accommodate those forces, defects appear quickly.
What determines the shape of a molecule?
The shape of a molecule is determined by electron-pair repulsion and bonding configurations, which push atoms into stable three-dimensional arrangements.
Electron pairs seek space, so atoms adopt shapes such as tetrahedral or trigonal planar, depending on their bonding patterns.
Hybridization explains how orbitals mix to give rise to these shapes, and that mixing influences bond strength and orientation.
Directional bonds often create stiffness, while less directional bonding allows movement.
Molecular forces also have a role.
Hydrogen bonding and van der Waals interactions impact how molecules pack, hence altering density and softening characteristics.
In practical terms, molecular shape becomes process behavior.
It influences how a material responds to bending, cutting, or heating on the shop floor.
How do bond angles and molecular forces influence material nature?
Bond angles and molecular forces influence material traits by controlling how easily a material can deform and how energy can move through it.
If geometry promotes tight packing and strong attraction, strength and melting temperature often increase.
However, increased rigidity can reduce toughness.
Conversely, flexible arrangements can improve impact resistance but reduce stiffness and load capacity.
Metals reflect similar logic at the lattice level.
Slip pathways allow deformation without fracture, while restricted movement promotes brittleness.
In manufacturing, these atomic decisions appear as springback, cracking, creep, or dimensional stability.
Geometry at the atomic level becomes performance at the component level.
How Do Atomic Bonds Translate into Mechanical Strength?
Atomic bonds translate into mechanical strength by defining the energy required to separate atoms and the ease with which they rearrange.
Strength, ductility, and fracture resistance are outcomes of the bonding type and the microstructure.
Mechanical strength includes tensile capacity, fatigue life, and resistance to fracture.
Each property develops from how atoms and grains interact under load.
Microstructure includes another layer.
Grain size, phase distribution, and defects influence how bonding effects are expressed at larger scales.
When forming or machining, these structural realities determine whether deformation is smooth or catastrophic.
Understanding this link prevents the trial-and-error process adjustments.
Types of Atomic Bonds in Engineering Materials
Atomic bonds in engineering materials are the essential bonds that define mechanical and electrical behavior.
The main types are ionic, covalent, metallic, and weak intermolecular bonds, and each influences performance differently.
Ionic bonds often create hard, high-melting materials that can be brittle.
Covalent bonds create strong directional networks that improve stiffness but limit plasticity.
Metallic bonding enables layers of atoms to slide while remaining connected.
This sliding ability supports ductility and formability.
Weak intermolecular forces predominate in many polymers.
They allow flexibility but can reduce high-temperature resistance.
Metallic Bonding
Metallic bonding is the shared electron structure in metals that allows atomic layers to move without breaking overall cohesion.
That mobility explains why metals can be bent, drawn, and formed.
In bending operations, ductility determines whether the outer radius cracks.
Slip planes in the crystal structure allow plastic deformation.
When working with CNC press brake machines, predictable metallic bonding as well as lattice behavior are essential for repeatable angles and minimal springback, which is why understanding the material science behind CNC press brake machines supports consistent production results
If metallic bonding is supported by proper grain orientation and processing, bending remains stable.
If not, cracking and inconsistent forming follow.
Covalent and Ionic Bonding
Covalent and ionic bonding dominate ceramics, semiconductors, and certain composites.
These bonds provide high stiffness and thermal stability.
However, directional bonding often limits plastic deformation.
Brittleness can become a concern in tensile loading.
In manufacturing, these materials are used in cutting tools, sensors, and high-temperature components.
Their success depends on matching bond-driven properties to process conditions.
How Do Crystal Structures Influence Industrial Materials?
The crystal structure is the repeating lattice arrangement of atoms in a solid that governs its deformation behavior.
In metals, the lattice controls how dislocations move, which defines ductility and strength.
Grain size and orientation add complexity.
Fine grains can increase strength, while certain orientations influence bend performance.
Manufacturing processes increase these effects.
Heat, strain, and cooling can alter phase balance and grain distribution.
Understanding lattice type helps predict forming limits and fracture risk.
Structure guides performance before production begins.
Body Centered Cubic vs Face Centered Cubic vs Hexagonal Close Packed
FCC, BCC, and HCP structures vary mainly in available slip planes, which influence ductility.
FCC metals are typically highly formable, while HCP metals can be less ductile at room temperature.
BCC metals frequently balance strength and ductility but may show temperature sensitivity.
Each structure suits different industrial roles.
Table: Crystal structure comparison
| Structure | Ductility | Strength | Temperature Sensitivity | Common Uses |
| FCC | High | Moderate | Low | Forming, wiring |
| BCC | Moderate | High | Higher | Structural components |
| HCP | Lower | Moderate to high | Moderate | Lightweight systems |
What Manufacturing Processes Rely on Molecular and Crystal Structure?
Manufacturing processes depend on how materials respond to stress and heat at the atomic and grain levels.
Deformation, cutting, and joining all interact with microstructure.
Material structure determines crack resistance, distortion, and surface finish quality.
Ignoring structure often leads to variable output.
Key processes controlled by atomic and crystal structure include:
- Sheet metal bending
- CNC machining
- Heat treatment
- Additive manufacturing
- Welding
CNC Press Brake Machines
A CNC press brake machine is a precision forming system that bends sheet metal using controlled force and programmable positioning.
Its performance depends heavily on ductility, work hardening, and grain structure.
Springback, cracking, and angle accuracy are all structural outcomes.
Choosing appropriate material and tooling improves consistency.
Controlled bending relies on both machine exactness and predictable lattice behavior.
Without that synergy, forming becomes unstable.
Servo Drive Systems in Automation
Motion control technology regulates position and velocity in automated equipment.
Precision motion depends on mechanical stiffness and structural stability.
Servo motor drives convert electrical signals into controlled motion.
If machine structures vibrate or deform unpredictably, accuracy suffers.
Understanding what is motion control in a practical context clarifies why material stiffness and damping matter in automated systems within a manufacturing framework.
Stable materials allow stable control loops.
Atomic structure indirectly influences automation reliability.
What Are the Advantages of Understanding Molecular Geometry in Manufacturing?
Understanding molecular geometry improves decision-making and reduces costly surprises.
It synchronizes material science with production goals.
Here are 6 main advantages:
- Improve material selection precision by linking structure to performance.
- Increase structural reliability by predicting failure modes.
- Reduce production waste by reducing the number of trial adjustments.
- Boost energy efficiency via optimizing thermal and friction behavior.
- Optimize machine calibration by anticipating springback and distortion.
- Support advanced innovation in alloys and composites.
What Are the Limitations of Applying Molecular-Level Knowledge in Industry?
Applying molecular knowledge can involve cost and complexity.
Laboratory-level insight does not always translate easily to production lines.
Here are 4 limitations:
- Increase research costs due to testing and analysis requirements.
- Require advanced equipment for microstructural validation.
- Complicated modeling when real materials contain defects.
- Limit rapid prototyping if variability is high.
How to Select the Right Material Based on Molecular Structure?
Selecting the right material requires assessing requirements, structure, compatibility, cost, and validation.
This process includes 5 essential steps.
- Define mechanical and environmental requirements, including load, temperature, and corrosion exposure.
- Analyze bonding type and crystal structure to predict ductility and strength.
- Evaluate manufacturing compatibility, including bending and machining responses.
- Consider cost and availability, including scrap rate and supply stability.
- Validate through testing and quality control to confirm real-world performance.
Material pricing varies widely by grade and form, and the cost must include both the purchase price and the processing impact.
Strategic validation confirms that molecular assumptions match the finished part’s behavior.
How Does Molecular Geometry Promote Innovation in Modern Manufacturing Technologies?
Molecular geometry drives innovation by embedding performance directly into structure.
Advanced alloys, semiconductors, and engineered surfaces rely on atomic-level design.
Instead of oversizing components, engineers now refine bonding and grid arrangement.
This strategy improves the strength-to-weight ratio and energy efficiency.
Contemporary automation, electronics, and green energy systems all depend on materials engineered at the atomic scale.
Structure becomes strategy in competitive manufacturing.
What Are Alternative Ways to Improve Material Strength Past Atomic Structure?
Strength can also be improved through design and processing.
These approaches complement atomic-level engineering.
Five common alternatives include:
- Increase geometric efficiency with ribs and reinforcements.
- Refine microstructure by controlled rolling or forging.
- Apply heat treatment to modify phase balance.
- Add surface treatments to enhance wear resistance.
- Improve joint design to reduce stress concentration.
Each method still interacts with the structure.
Processing modifies how atoms and grains arrange themselves.
Conclusion
Atomic bonds are the foundation of industrial strength.
Molecular geometry shapes material properties, and crystal structure translates those properties into performance under stress.
When manufacturers understand this chain from atom to assembly, they reduce guesswork and improve reliability.
Material science becomes a practical tool rather than a conceptual theory.
From bending with press brake systems to controlling motion with servo drives, structure affects outcomes at every stage.
Industrial success commences long before the first machine cycle, at the atomic level.